
Currently, predominantly axial back pain in patients without a history of spine surgery is not an FDA-approved indication for SCS.Ī 53-year-old-female with a medical history of diabetes mellitus type 2, LBP, and lumbar radiculopathy presented to the clinic in December 2019 for LBP with intermittent radiation to the left lateral thigh. Shealy, they continue to rely on this theory.Ĭurrently, Food and Drug Administration (FDA)-approved indications for spinal cord stimulation include failed back surgery syndrome or postlaminectomy pain, cervical and lumbar radiculopathy, complex regional pain syndrome types I and II, diabetic peripheral neuropathy, arachnoiditis, epidural fibrosis, post-thoracotomy pain, and postherpetic neuralgia. Although there have been considerable improvements to the technology used in SCS today compared to those used by Dr. 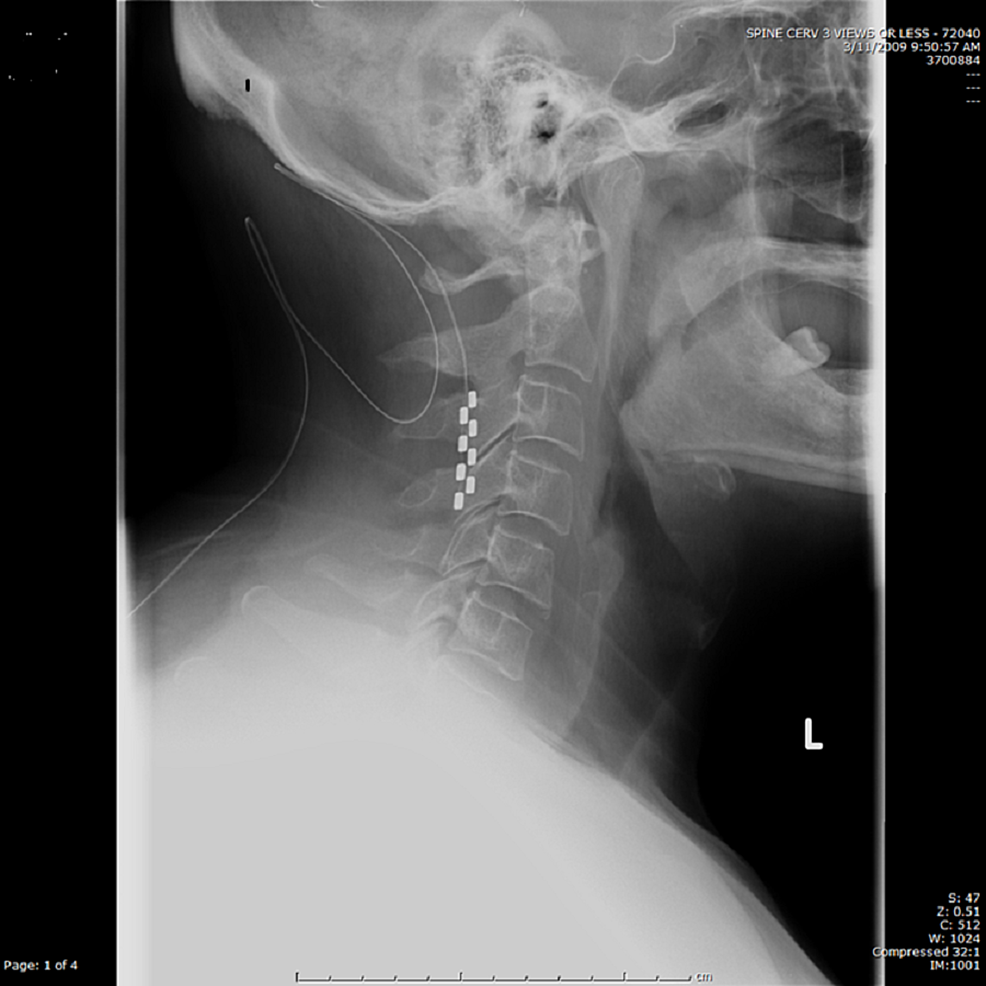
They postulated that increased stimulation of non-nociceptive fibers of the dorsal columns would reduce or block signal transduction of smaller nociceptive fibers to supraspinal targets. The first spinal cord stimulators (SCS) were designed based on the gate control theory initially proposed by Melzack and Wall in 1965. Neuromodulation has been used in medicine for over 50 years, with the first dorsal column stimulator implant performed in 1967 by Dr. In the United States alone, the economic impact of workdays lost secondary to LBP has been estimated at 100-200 billion USD each year. 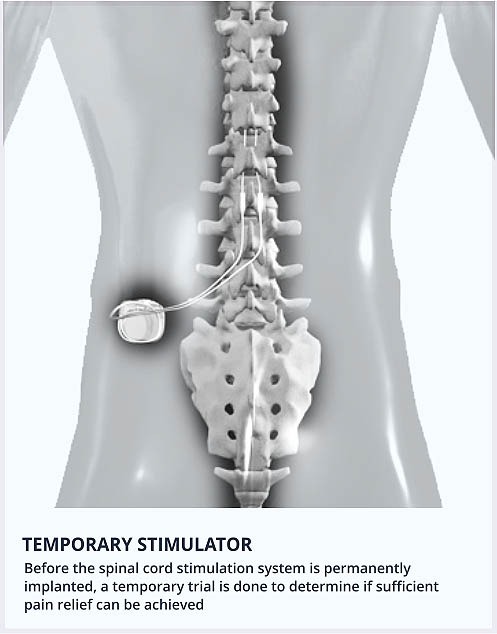
Chronic pain, especially low back pain (LBP), is one of the leading causes of physical activity limitation, absence from work, and disability globally. The prevalence of LBP in 2017 was estimated to be 7.5% globally or approximately 577 million people, with the lifetime prevalence approaching 60-70%.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
